Electronic charge flow through molecular scaffolds
Electron transfer is one of the most fundamental and ubiquitous chemical phenomena in nature, driving processes necessary for life such as photosynthesis and respiration. From a technological standpoint, understanding the flow of electronic charge through organic molecules is the basis for developing biosensing and molecular electronics applications. Further, efficient transport of electronic charge through organic and inorganic scaffolds is a critical requirement in material design for solar energy harvesting or photocatalytic applications. At TIFR, our group has developed a versatile theoretical framework capable of describing electron transport through molecules in a wide variety of natural and synthetic environments.
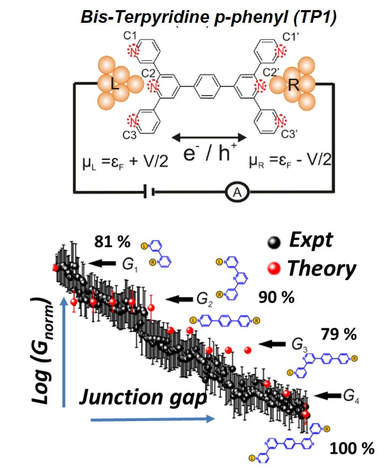
Molecular Breadboard CircuitsIn 1974, Aviram and Ratner proposed the concept of a molecular rectifier, thereby creating the field of molecular electronics.The measurement of single molecule conductance through mechanically controlled or STM break junction techniques (MCBJ/STM-BJ) offer the potential to examine molecules as electronic circuit elements (e.g. wires or switches or diodes). At present, understanding the electronic features of molecular components embedded within a macromolecular scaffold is an unsolved fundamental challenge in the field of molecular electronics. My group has made progress in this direction by demonstrating that single organic molecules can function as scaffolds to efficiently build rewire a multitude of embedded microscopic circuits with distinct conductance features.
Through theoretical studies of the conductance of a bis-terpyridine p-phenyl (TP1) molecular junction (Figure on the right) we translated the concept of a breadboard circuit, well-known in conventional macroscopic electronics, to the molecular scale. As such, previous state-of-the-art studies have only been able to create and study 2 to 3 circuits within rigid molecular scaffolds. In comparison, we showed that the TP1 molecular scaffold offers 61 single and multi-terminal circuits embedded within a flexible scaffold! A record 4 well resolved conductance states, spanning 5 orders of magnitude were experimentally resolved for the TP1 molecular junction by our collaborators |
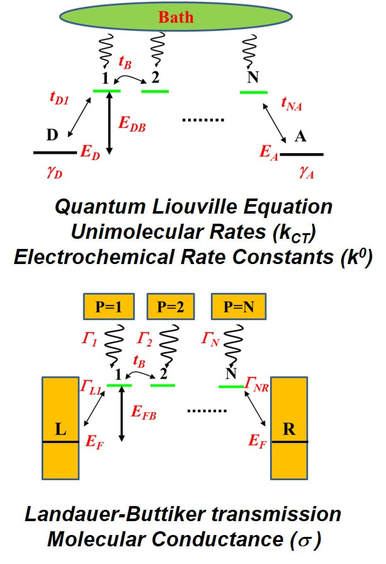
The relationship between molecular conductance and charge transfer ratesIn molecular electronics, the ability of organic molecules to transmit electronic charge is a fundamental property of interest. The charge transmitting ability of molecules can be captured experimentally by two complementary measures: 1) molecular conductance from nanoscale conducting junction experiments and 2) charge transfer rates from time resolved spectroscopy and electrochemistry experiments. Should these two measures correlate? This question is highly relevant for measurements of charge transport efficiencies across nanoscale molecular assemblies and for creating a consistent physical interpretation of data from different types of charge transport measurements.
Our group has established a theoretical framework (Figure on the left) which, independent of the underlying charge transfer mechanism, enables a comparison of the unimolecular charge transfer rate and molecular conductance through a minimal set of transferable parameters. Through the newly established framework, we have demonstrated that differences in the barrier heights for charge transport, and in bath-induced decoherence effects can have a profound effect on the correlation of the two different charge transport measures even when the charge transfer mechanism is the same during the measurements. Along with our collaborators we have showed that non-linear rate-conductance relationships for a molecule would follow a power law when described as a function of molecular length. The newly developed theoretical framework helps rationalize puzzling nonlinear relationships between experimental electrochemical charge transfer rate constants and molecular conductance measured for a variety of molecules, in different regimes of charge transport by our collaborators. |